Introduction
Carpal tunnel syndrome (CTS) is the most common cutaneous entrapment neuropathy, affecting 3–6 per cent of the general population.1 Carpal tunnel release is routinely carried out in cases of CTS and it is among the most common wrist and hand surgical procedures. Various anatomical anomalies within the wrist and carpal tunnel have been reported in the literature. Familiarity with aberrant tendons, nerves and/or muscles is vital to avoid iatrogenic injury during an otherwise routine operation.
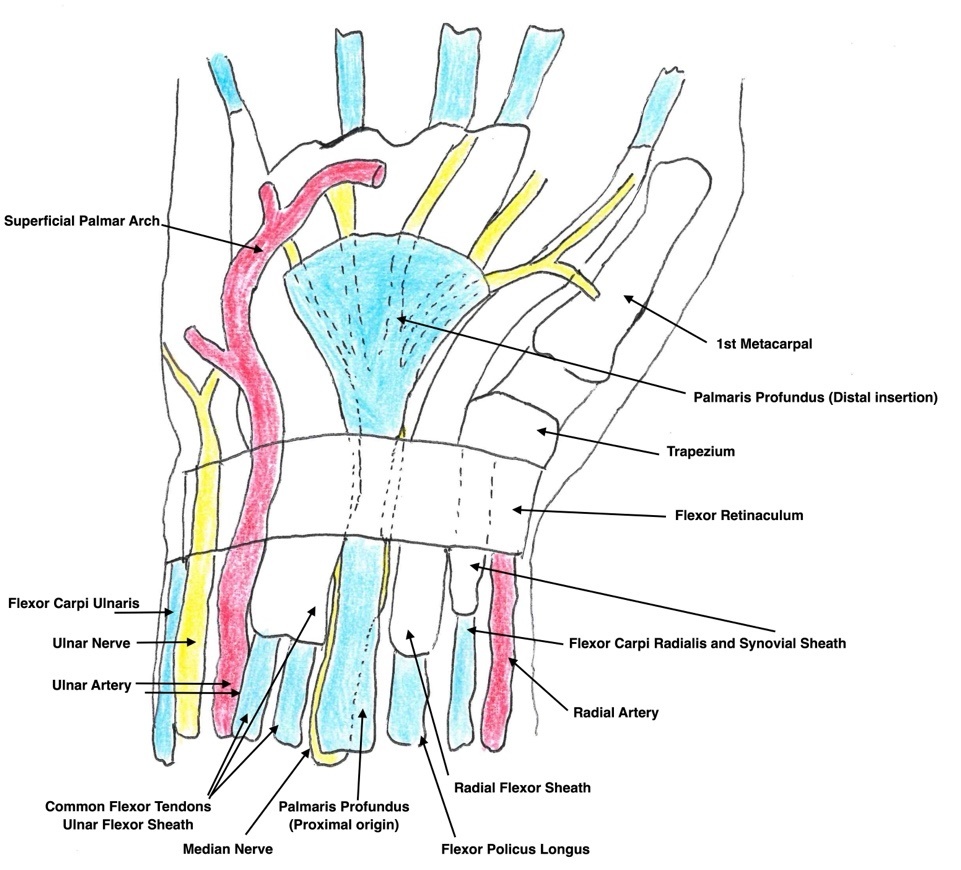
The palmaris profundus (PP) is an anomalous muscle of the hand and wrist (see Figure 1). It was first described by Frohse and Frankel in 1908.2 Reimann and colleagues in 1944 reported an exceedingly low incidence of 0.125 per cent3 from initial cadaveric dissections. This muscle is often encountered only during open carpal tunnel release. Described as musculus comitans nervi mediani, the PP is often found within the same fascial sheath as the median nerve.4 Most data arise from a series of case reports. Pirola and colleagues have documented three PP subtypes based on origin: Type I arises from the radius; Type II arises from the fascia of the flexor digitorum superficialis; and Type III arises from the ulna.5 All subtypes report a tendinous insertion to the palmar aponeurosis at the level of the wrist.
Carpal tunnel syndrome is exacerbated by factors which may increase the pressure within the enclosed carpal tunnel.8 The presence of an additional PP muscle, often investing into the same fascial sheath as the median nerve, is a discrete risk factor in CTS.1,4,5 We provide, to the best of our knowledge, the first published systematic review assessing the incidence, variations and surgical implications of the PP on CTS. We aim to shed light on a rare anatomical anomaly with the propensity to complicate an otherwise routine surgical procedure.
Methods
Search strategy
A systematic literature search was conducted on March 22, 2025 across four electronic databases: MEDLINE, Embase, PubMed and Cochrane Library. The objective was to identify studies describing CTS caused by the PP tendon. The search strategy incorporated a combination of Medical Subject Headings (MeSH) terms and keywords to ensure comprehensive coverage. Boolean operations (AND, OR) were used to appropriately combine search terms and refine results. The full search strategy for each database is provided in Tables 3–6 of Supplementary Material 1.
Eligibility criteria
Inclusion criteria:
-
full-text studies describing CTS associated with a PP tendon
-
published in English.
Exclusion criteria:
-
studies describing CTS secondary to anomalous palmaris longus muscles or other causes of median nerve compression not involving the PP tendon
-
studies employing the wrong study design, including narrative or scoping reviews, cadaveric or imaging-only anatomical studies without clinical correlation and conference abstracts or non-full-text studies
-
non-English language studies.
Study selection
Title and abstract screening, followed by full-text review, was conducted independently by two independent reviewers (RK and GJC) using Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia). Discrepancies were resolved by consensus, with involvement of a third reviewer (AT) if required. The study selection process was conducted in accordance with PRISMA guidelines.9 This systematic review was not registered with PROSPERO.
Risk of bias assessment
Risk of bias was assessed using the Joanna Briggs Institute Critical Appraisal Checklist for Case Reports,10 as all included studies were case reports or case series. No studies were excluded based on the risk of bias assessment. A summary of the appraisal is provided in Table 1 of Supplementary material 1.
Data extraction
Data were systematically extracted using a structured Microsoft Excel (version 16.106.1, Microsoft Corporation, Redmond, Washington, USA) spreadsheet to ensure consistency and accuracy. Extracted variables included:
-
study characteristics—title, authors, year of publication, country, study design and sample size
-
patient demographics—age, sex and comorbidities
-
diagnostic details—examination findings, conduction studies, imaging, carpal tunnel syndrome instrument
-
surgical management—surgical approach, intraoperative findings, complications
-
postoperative outcomes.
Results
Search results
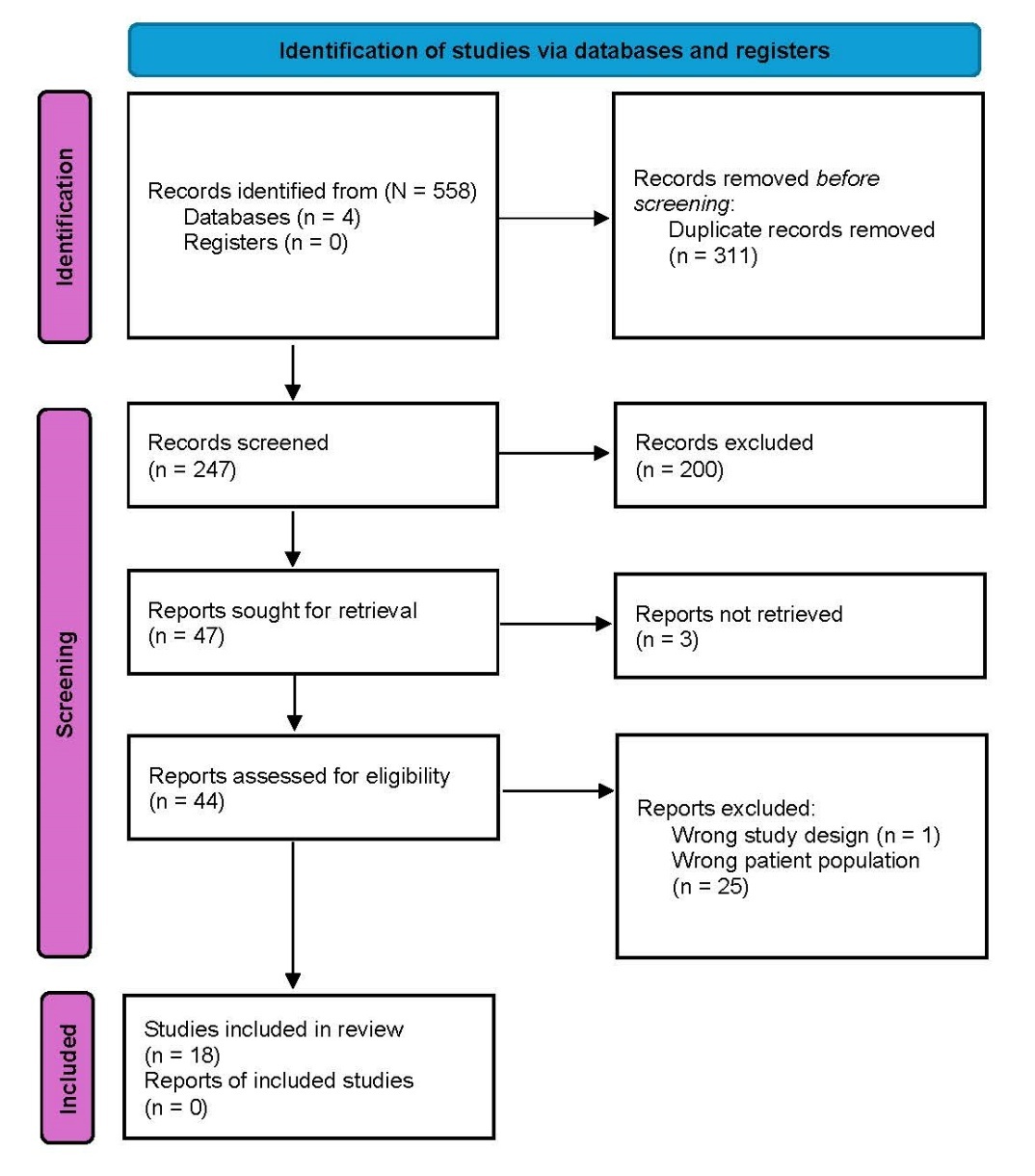
A total of 558 articles were identified through searches of MEDLINE, Embase, Cochrane Library and PubMed (Figure 2). After removing 311 duplicates, 247 studies remained for title and abstract screening. Of these, 200 studies were excluded, leaving 47 for full-text assessment. Three articles could not be retrieved, resulting in 44 full-text articles reviewed for eligibility. After full-text screening, 26 studies were excluded, leaving 18 studies included in the final systematic review.1,5,11–26
Study population
A total of 21 patients were included in this cohort (see Table 1), with a median age of 65.0 years. Eleven patients (52.4%) were male1,5,15,18–21,23,26 and nine (42.9%) were female5,11–14,16,22,24,25; sex was not reported for one patient (4.7%).17 The condition more frequently involved the right side (76.2%, n = 16)1,5,11,14,16–18,20–26 compared to the left (23.8%, n = 5).12,13,15,19,26
Preoperative investigations varied across the cohort. Nerve conduction studies were performed in 14 patients (66.7%),1,11,13–20,22,23,25 while electromyography was conducted in only three cases (14.3%).12,26 Ultrasound11,20 and MRI5,11,21 were each used in three patients (14.3%). Use of the carpal tunnel syndrome instrument was reported in a single patient (4.8%).1
Clinical presentation
The most reported symptom (see Table 2) was paraesthesia (90.5%, n = 19),1,11–20,22–26 followed by nocturnal brachialgia (42.9%, n = 9)11,13,15–17,22–24 and functional limitation (19.0%, n = 4).11,13,18,22 Positive Tinel’s and Phalen’s signs were recorded in nine (33.3%)11,13–15,17,19,20,22,25 and five patients (23.8%)1,11,14,19,25 respectively. Thenar atrophy was observed in three patients (14.3%).14,16,24
Surgical management and outcomes
The predominant surgical approach (see Table 3) was open decompression (95.2%, n = 20),1,5,11–26 with only one case involving a combined endoscopic and open technique (4.8%).16 Intraoperatively, a bifid median nerve was identified in seven patients (33.3%).1,5,12,16,20,23 Index operative intervention on the PP muscle included resection or release in 20 patients (95.2%).1,5,11–26 One patient (4.8%)20 had no surgical intervention on the PP and received a steroid injection to good effect. Visualised compression of the median nerve was noted in 14 patients (66.7%).1,5,12,14–17,19–22,24,25 The PP was most found to arise from the flexor digitorum superficialis fascia (38.1%, n = 8),5,10,19,20,26 followed by other carpal tunnel origins in two cases (9.5%)15,21 and the ulna head in one case (4.8%).24 Insertion into the palmaris aponeurosis was reported in 81.0 per cent of cases (n = 17).1,5,11,15–20,22,24–26 The remaining four insertions were not reported on.12–14,23
Complications were reported in three patients (14.3%),14,15,24 and reoperations were required in three cases (14.3%).14,15,24 Overall, complete symptom resolution following index surgery was achieved in 18 patients (85.7%).1,5,11–13,16–23,25,26
Postoperative complications
Three studies reported postoperative complications (Table 4).14,15,24 These included hand pain, symptom recurrence and compartment syndrome. All cases were managed with further intervention on the PP, with complete symptom resolution documented in each case.
Discussion
Palmaris profundus is an exceedingly rare variant of forearm musculature observed within the carpal tunnel. First described by Frohse and Frankel2 in 1908, its cadaveric incidence is exceedingly low, reported in 1 in 800 limbs or a rate of 0.125 per cent. Consequently, current understanding of the PP arises from isolated case reports. It is often intimately associated with the median nerve, found within the same fascial sheath,4 and is thus also referred to as musculus comitans nervi mediani.
The PP exhibits considerable anatomical variability. Priola and colleagues described three principal subtypes according to the muscle’s proximal attachment: Type I from the radial shaft, Type II from the fascia of flexor digitorum superficialis and Type III from the ulna shaft.5 Other less common origins have also been reported, including the medial epicondyle or a slip from the palmaris longus.5 Insertion is far more consistent: in all reported cases the PP tendon fans out into the deep palmar aponeurosis at the level of the wrist. Notably, the PP tendon typically lies volar and radial to the median nerve. Rare descriptions, crossing from the radial to ulna shaft across the nerve, exist.27 No single pattern of innervation or vascular supply has been established, and the muscle appears to have no discernible function. In agreement with prior authors, we regard the PP as essentially a vestigial, developmental remnant of the anterior forearm musculature.5,8–14
From an embryological standpoint, the PP may represent a deep part of the ancestral palmaris musculature or an aberrant slip of the flexor digitorum superficialis complex.28 In any case, it serves no critical function; most patients report no post-resection functional deficit. This compares to other anomalous structures, such as an aberrant lumbrical or nerve, which may have functional consequences.29
This close anatomical relationship makes the PP a plausible compressive lesion in the aetiology of CTS. An extra tendon coursing through the carpal tunnel reduces the available space for the nerve. The PP is best viewed as a potential anatomical risk factor—a volume-occupying structure that can precipitate or exacerbate nerve entrapment.13,15,18
By analogy with other secondary causes of CTS, such as tendonitis, synovitis or intracarpal oedema,30,31 a PP in the carpal tunnel plausibly increases compartment pressure and predisposes to CTS. Although no experimental study quantifies this effect, it is intuitive that any added, volume-occupying structure in the tunnel may further compress the median nerve.13,15,18 Our review supports this idea, with 66.7 per cent of reported cases documenting intraoperative evidence of median nerve compression by the PP. Other reports explicitly note that the anomalous tendon was enveloping or displacing the nerve.15,18 It is challenging to attribute a symptomatic role of the PP to CTS. McClelland and colleagues emphasise that, although the PP is non-functional, the tendon is implicated in CTS.16 Likewise, the case report by Pezas and Jose states that a PP, given its course under the flexor retinaculum, reduces the space available within the carpal tunnel for the median nerve to traverse.18 Taken together, the literature and our findings support the concept of the PP as a potential but rare causative factor in CTS.
This review of 21 patients demonstrated that cases were predominantly right-sided (76.2%), inline with previous reviews noting a strong right-hand preponderance (on the order of 6:1).32 Nearly all patients underwent open carpal tunnel release, reflecting standard surgical practice. The single endoscopic case was converted to an open procedure following technical resistance upon inadvertent encountering of the intracarpal PP.
Resection of the PP was performed in almost every case (95.2%). Most patients (85.7%) reported complete or near complete paraesthesia resolution reflecting significant post-surgical symptom relief. This aligns with prior case reports in that release or resection of the PP tendon led to immediate and lasting symptom improvement.14,32 Lyrtzis and colleagues14 note complete resolution of symptoms and return to normal activities in their revision case report. Here, the authors brought an open carpal tunnel release back to the operating theatre following persistent post-release symptoms. In this second operation, they resected an initially missed PP tendon. The authors attribute symptom relief to this revised resection.
Only two patients had early complications (wound dehiscence and haematoma). Three patients reported postoperative complications, all requiring repeat operations (symptom persistence in two cases and concerns for compartment syndrome of the hand in one). All three cases noted incidental and missed PP, requiring revision release. This highlights that when the PP is correctly identified and excised, the prognosis is excellent. Overlooking it may necessitate reintervention.14
In cadaveric and imaging studies, the bifid median nerve has been reported to have an incidence of 2–26 per cent.33–36 The review demonstrated that approximately one-third of cases had an associated bifid median nerve (with or without a persistent median artery). The co-occurrence of PP and other nerve anomalies has been reported in previous instances. McClelland and colleagues reported a persistent median artery and bifid nerve in the same patient with a PP.16 Some authors have even documented bilateral PP in association with bifid nerves as the reason for failed carpal tunnel release.14 A previous meta-analysis has demonstrated that a 50 per cent greater incidence of bifid median nerves were found in patients with CTS than patients without CTS. A bifid median nerve increases the mean cross-sectional area of the wrist by 1.5 mm.2,37 This shows that anatomically, the bifid median nerve is a risk factor for CTS. This mirrors our findings, with 33.3 per cent of patients having a bifid median nerve discovered during carpal tunnel surgery. While our numbers are small, this suggests that encountering a PP should prompt careful inspection for other anomalies.
A major challenge highlighted by our review is limitations in establishing preoperative diagnosis. In almost all cases, a PP was not correctly identified until the time of surgery. Physical examination in the context of CTS does little to raise suspicion of aberrant structures, including the PP.38 Furthermore, preoperative imaging is seldom carried out, in keeping with current surgical practice. Our findings support this, with only four out of 21 patients having targeted imaging (ultrasound or MRI).5,11,20,21 Only one of these four studies identified the PP.20 Notably, this patient was referred for sonographic guided corticosteroid injections of the carpal tunnel. At the time of this intervention, the anomalous PP was identified via ultrasound.20 None of these scans definitively reported a PP. The literature remains undecided on the utility of diagnostic adjuncts in assessing for aberrant structures and their significant on CTS.39,40 Flores and colleagues15 have suggested that ultrasound may be valuable in the preoperative planning for carpal tunnel release, especially in revision cases. Accordingly, while we do not advocate routine imaging for uncomplicated CTS, we suggest that in atypical presentations or when there are recurrent symptoms, targeted imaging including high-resolution ultrasound or MRI should be considered. This could potentially reveal an occult PP and guide surgical planning. Indeed, protocols for peripheral nerve imaging are growing, and CTS cases with failed initial decompression may particularly benefit from such evaluation.41,42
Given these findings, heightened surgical awareness of the PP is important. Surgeons performing carpal tunnel release should be cognisant of the variant tendon, especially if the tunnel seems unusually tight intraoperatively or if a tendon-like structure is noted. Intraoperative decision-making should favour release or resection of the PP when identified. Excision is straightforward; the tendon is visible once the retinaculum is released. This adds minimal operative time and markedly reduces the risk of residual compression.13,18,21 In fact, leaving the PP intact is ill-advised, as per Lyrtzis and colleagues’ experience discussed earlier.14 Given this, our recommendation, consistent with the data in this review, is to excise any PP encountered. The PP accordingly should be treated like any other compressing structure. The minimal added risk of excision, combined with the high likelihood of symptom resolution,14,32 strongly supports this strategy.
Several limitations of our review and underlying evidence warrant mention. First, PP-associated CTS is so rare that only case reports and small series exist. There are no controlled studies and no systematic anatomical surveys that assessed the PP in the context of CTS. Publication bias is likely, as unusual or surgical failure cases and positive outcomes are more likely to be reported. Reimann’s classic dissection in 19443 provides a crude incidence estimate, but no recent large cadaver series has updated this figure. Consequently, the true prevalence of the PP (even among CTS patients) is unknown. Moreover, because a PP is usually asymptomatic until found intraoperatively, its identification is largely incidental. Proving causality is therefore exceedingly difficult. One cannot ethically randomise or compare groups with and without a PP. As a result, all data must be interpreted cautiously. We have mitigated this by systematically gathering all reported cases, but the level of evidence remains low. Finally, imaging protocols varied widely, if used at all. We cannot quantitatively assess the sensitivity of ultrasound or MRI for detecting a PP. This heterogeneity highlights the absence of standardised diagnostic pathways.
Our findings suggest several directions for improving knowledge and management of a PP. A prospective surgical registry of carpal tunnel release cases would be valuable. Simply recording whether a PP, or any other anomaly, was visualised would aid in estimating its incidence. For patients with persistent or recurrent CTS after surgery, incorporating dedicated imaging protocols should be studied: for example, routinely performing ultrasound to assess for residual compressive structures (such as a PP) might shorten time to diagnosis. Ongoing education of plastic or hand surgeons and radiologists is crucial; the PP should be included in teaching materials about CTS variants. Finally, basic anatomical research might clarify whether there are genetic or developmental factors predisposing some individuals to having a PP, perhaps related to the high association with bifid nerves. In any event, until more data are available, awareness is key. Surgeons should keep the PP in mind when additional or unexpected structures are visualised within the carpal tunnel, and should document and report these cases.
Conclusion
The PP is a rare but clinically significant anomaly in the context of CTS. Although it accounts for a small proportion of CTS cases, its presence can complicate an otherwise routine operation. Our systematic review indicates that vigilance for this variant, combined with prompt excision when found, yields excellent outcomes. Until larger data emerge, the practical guidance is clear: in cases of CTS (especially atypical), consider the possibility of a PP and, if encountered intraoperatively, resection increases the chances of full decompression and subsequent symptom relief and reduces clinical recurrence.
Conflict of interest
The authors have no conflicts of interest to disclose.
Funding declaration
The authors received no financial support for the research, authorship, and/or publication of this article.
Revised: November 4, 2025 AEST