The surgical landscape for the treatment of upper limb spasticity has remained static until recently. Arthrodeses, soft tissue releases and tendon transfers have been the surgical mainstays in patients recalcitrant to non-operative measures for good reasons—when used in the right patient, these procedures yield consistent and satisfactory outcomes.
Increased tone, however, results in muscle imbalance, which, particularly when combined with paresis of antagonistic muscles, leads to joint contractures and deformity. Traditional modalities only address contracture, imbalance and deformity without treating the underlying spasticity itself. Thus, surgery was often reserved for those with more advanced disease or severe disability. Patients with early disease or those with spasticity alone were restricted to medication or regular, temporary botulinum toxin injections.
Over the past decade there have been steady advances in nerve-based techniques that aim to target spasticity directly. In particular, the use of hyperselective neurectomy (HSN) has become increasingly widespread. We have been incorporating HSN in our algorithm for the management of spasticity for the past few years with pleasing outcomes. This technique and its place in the surgeon’s armamentarium when treating spasticity is the focus of this editorial.
First, a brief recap on the stretch-reflex arc that underpins spasticity. When a muscle is stretched, type 1A sensory fibres in the muscle spindle relay this information to the spinal cord. Here, alpha motor neurones are activated via a monosynaptic reflex, causing extrafusal muscle fibres to generate force and resist that stretch. Simultaneously, antagonistic muscles are relaxed via a polysynaptic reflex to also prevent contraction, via a mechanism known as ‘reciprocal inhibition’. In non-pathological states, passive stretch does not result in activation of the stretch-reflex arc, due to dampening via descending fibres. However, following an insult to the upper motor neurons (such as in cerebral palsy, traumatic brain injury, cerebrovascular accidents or spinal cord injury), these descending inhibitory mechanisms are impaired. There is less inhibition, resulting in increased resistance to stretch—the degree of this resistance is dependent on the velocity at which the muscle is being stretched.
The technique of HSN has been championed over the last decade by a small but growing group of surgeons. In particular, Caroline LeClercq at Institut de la Main in Paris, France, has spoken and published extensively on the technique and its relevant neuroanatomy.1–4 The concept of dividing peripheral nerves to reduce spasticity, however, is not novel. It was initially described in 1913 by the German anatomist and surgeon Adolf Stoffel.5 His other innovations include the nerve stimulator and early descriptions of many of the nerve transfers that we employ today.6 Unfortunately, likely due to the political tensions of the day, his ideas were not adopted or revisited, until Brunelli and son reported on a similar technique in 1983.7
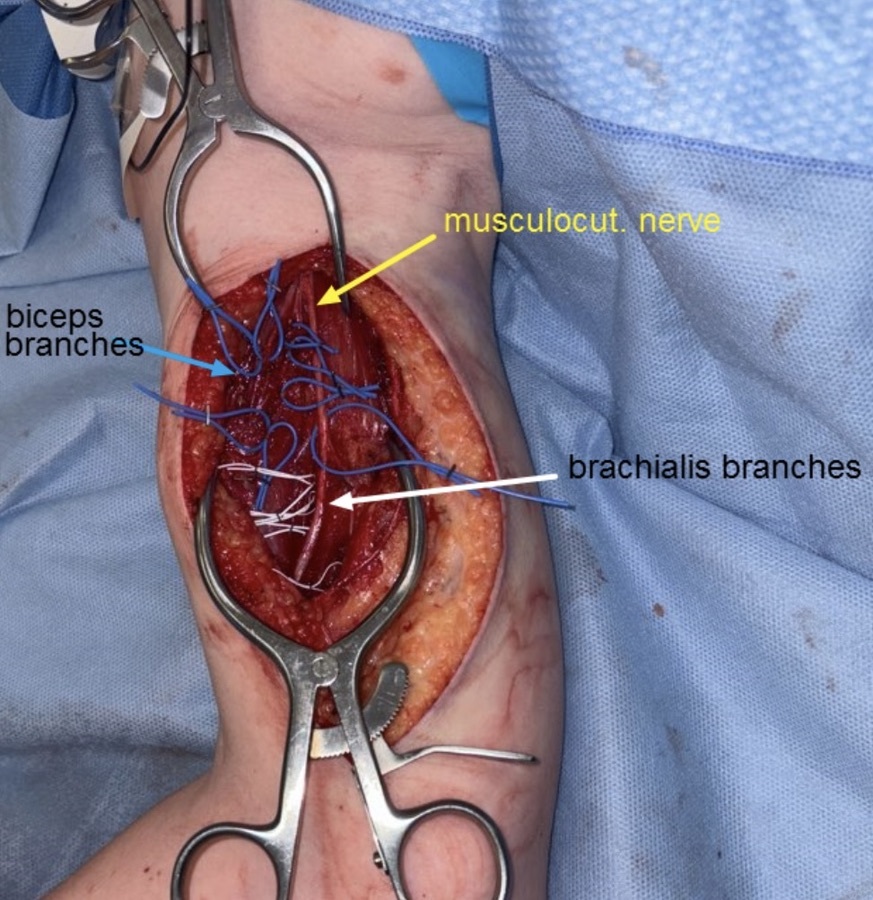
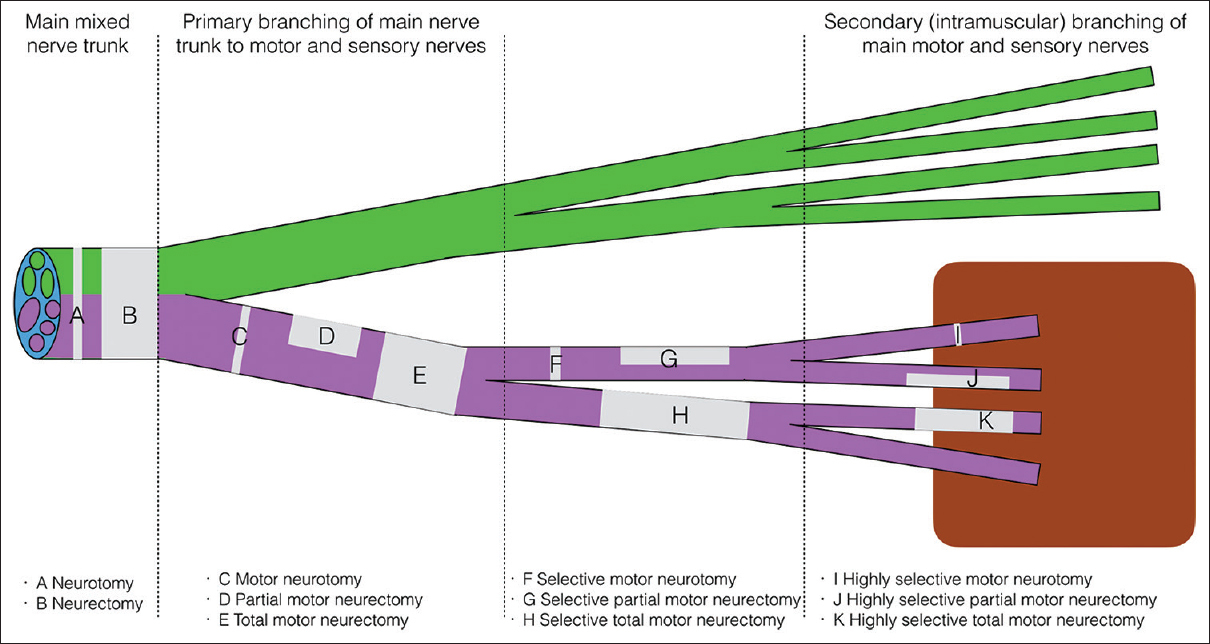
The aim of HSN is to reduce spasticity without affecting power, and it involves dissection of all secondary motor nerve branches to a spastic muscle, with division of 70–80 per cent of these (Figure 1). The level of the neurectomy is important—identification of the secondary motor branches requires intramuscular dissection until the ramifying nerve is seen to enter the muscle fibre (Figure 2). It is hypothesised that the remaining efferent motor fibres can collateralise to adjacent denervated muscle post neurectomy via a process known as adoption, creating a larger motor unit and restoring full power over time, while the afferent fibres driving spasticity cannot, thus permanently diminishing spasticity. This can be thought of as permanent botulinum toxin, but better—HSN reduces spasticity but maintains power, while botulinum toxin reduces both.
The mechanism of action is certainly not definite and, while the above is a convenient explanation, the reduction in spasticity is also likely to be the result of ‘synaptic stripping’. When peripheral nerves are completely transected and repaired, the stretch-reflex arc is absent even if full motor function returns—Alvarez found that this is due to a loss of central (spinal cord) synapses on the proprioceptive arc.8 This reduces spasticity effectively and some surgeons are beginning to explore a simple ‘cut and repair’ technique on peripheral nerves (also termed ‘reset neurorrhaphy’ or ‘section and repair’) that are driving problematic spasticity (such as the motor branch of the ulnar nerve in Guyon’s canal or the C7 nerve root). Contralateral C7 nerve transfer has also been used with encouraging results, predominantly in China.9 These more proximal techniques are in their infancy and, although genuinely interesting and offering promise, are not yet widely used. It is exciting to consider, however, that if the synaptic stripping hypothesis is true, one can perform peripheral limb surgery and effect a morphological change at the anterior horn of the spinal cord—in other words, we are moving our treatment effect closer to the source of the problem, while operating in the relative safety of the periphery.
Because HSN targets spasticity and spasticity precedes contractures, we are now operating on a cohort of patients who are either younger (in the case of cerebral palsy) or earlier in the disease trajectory (in other upper motor neurone syndromes). Referral patterns between rehabilitation physicians and surgeons have had to adapt—the essential change is that patients whose spasticity is well-managed via botulinum injections are now being seen earlier as potential candidates for HSN, rather than later when the effects diminish and contractures emerge. We are fortunate in our institutions to meet these patients in a multidisciplinary setting that includes physicians, therapists and surgeons.
Now that there are more tools in the armamentarium, one is better able to apply the correct tool for the job. We use HSN for spasticity; tenotomies, tendon transfers or tendon lengthening for muscle imbalance; and releases, stabilisations and fusions for end-stage joint contracture. It is, and always has been, an imperative clinical step to differentiate spasticity (velocity dependent tone) from contracture (restricted range of motion). Abnormal movement profiles (such as dystonia) need to be identified—and although not a contraindication to surgery, they typically render outcomes much less reliable. It is important to clarify the aims of the intervention carefully with the patient and their family. For highly functional patients, this might relate to improvements in function or cosmesis; for those with less volitional control, the primary aim may be hygiene or ease of dressing.
We perform multi-level surgery (shoulder down to fingers) where possible for logistical efficiency, and often combine techniques. For example, a common surgical sequence involves HSN to reduce elbow flexor spasticity, combined with a tendon transfer to balance the wrist, fractional lengthening of the long finger flexors, and a pronator teres release to improve supination.
In conclusion, nerve-based procedures such as HSN are effective and have renewed interest in the surgical treatment of spasticity. They do not supplant established techniques, but rather expand the surgical armamentarium, enabling targeted treatment of spasticity and contracture with the appropriate modality. Other nerve-based approaches, including C7 transfer or ‘cut-and-repair’ strategies, are promising but require further validation before routine use.
Patient consent
Patients/guardians have given informed consent to the publication of images and/or data.
Conflict of interest
The authors have no conflicts of interest to disclose.
Funding declaration
The authors received no financial support for the research, authorship, and/or publication of this article.