Introduction
Meningococcal disease carries a mortality rate of around 10 per cent in developed countries and of those who survive septic shock with purpura fulminans, eight per cent require digital and/or limb amputation.1,2
Neisseria meningitidis colonises the nasopharynx of approximately 10 per cent of the population.3 The sequelae of invasive infection are often devastating, with profound septic shock and disseminated intravascular coagulation (DIC) resulting in multiorgan microvascular thrombosis, tissue infarction and extravasation of blood. In the dermis, this manifests as purpura fulminans.4
The coexisting muscle and skin necrosis, once debrided, results in more complex defects than most full-thickness burns or necrotising fasciitis, which frequently spare a ‘graftable’ wound bed. The high amputation rate becomes apparent when presented with large ‘ungraftable’ wounds and limited available autologous tissue reconstructive options. Despite the following case being a rare entity, complex wound coverage remains a common challenge for the reconstructive surgeon.
Expanding experience with NovoSorb Biodegradable Temporising Matrix™ (BTM, PolyNovo Biomaterials Pty Ltd, Port Melbourne, Victoria, Australia) for complex wounds encouraged its use in the following case, which required reconstruction of extensive soft-tissue defects with exposed bone resulting from the sequelae of meningococcal B infection with septic shock and DIC.
Case report
A 17-year-old male presented to hospital, febrile (40°C), tachycardic (150 bpm) and with small, non-blanching spots on his flank. In the emergency department, the spots rapidly evolved into a purpuric rash and he developed vasopressor-dependent septic shock with severe DIC. Mottling of his extremities, accompanied by elevated serum creatinine kinase, mandated fasciotomies to all limbs.
In ICU he required intubation, coagulopathic correction, vasopressor support and haemodialysis. Once stable on day 13, he was transferred to the Adult Burn Service at the Royal Adelaide Hospital (RAH) for wound management constituting 19 per cent total body surface area (TBSA). Debridement of infarcted musculature and 19 per cent TBSA purpura fulminans resulted in near-circumferential soft-tissue loss to both lower limbs also extending over the bilateral thighs, forearms, abdomen and left hand.
The right leg wound contained exposed tibia and fibula denuded of periosteum, tendons lacking paratenon and the cut surface of muscle. Additionally, there was insensibility to both soles of the feet. Even in the absence of this latter finding, such circumstances would often result in bilateral below-knee amputation, because such defects would require multiple free flaps for coverage. In this case, compromised lower limb vasculature discouraged flap anastomosis below the knee, both anterolateral thigh flap donor sites were involved in the purpuric necrosis of the thighs and the use of latissimus dorsi flaps would impede subsequent aided mobilisation/wheelchair use.
It was decided to embark upon a reconstruction to preserve the legs that provided immediate coverage, avoided early use of skin graft/flap resources, gave time for potential re-innervation to the soles and allowed the patient to make his own choices regarding amputation at a later stage. Once satisfied with the debridement, all wounds were covered with BTM (day 14 for forearms/left hand/right thigh and day 29 for left thigh and leg). The BTM on the forearms, left hand and right thigh was delaminated and autografted on day 49 (35-day-old BTM) and on the left thigh and leg it was delaminated and autografted on day 75 (46-day-old BTM) with complete skin graft take.
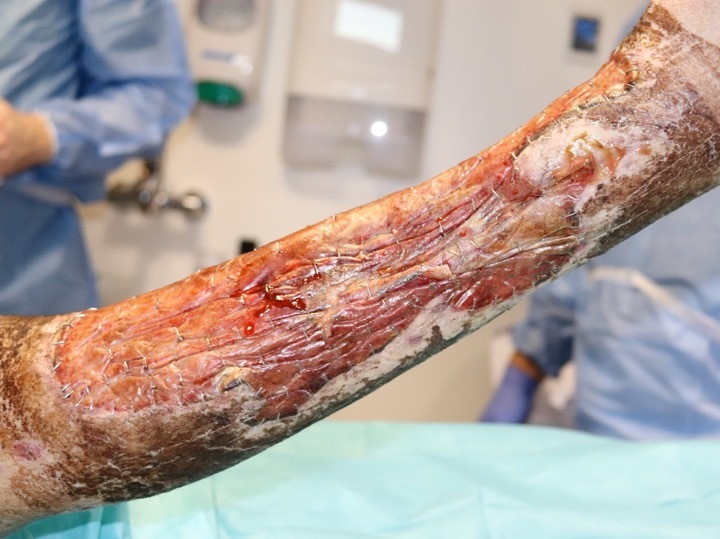
Debridement of the right leg wounds took longer and the BTM was applied on day 40. To facilitate integration, two millimetre burr-holes were drilled through exposed tibial and fibular cortices prior to application (Figure 1). On day 75, once the right leg BTM had integrated (35-day-old BTM; Figure 2), it was delaminated and grafted.
The leg grafts demonstrated 95 per cent take at first dressing change on day 81, with spontaneous healing of the remaining areas, and allowed mobilisation in ankle-stabilising orthoses on day 82. The patient’s admission was complicated by carbapenem-resistant Pseudomonas aeruginosa wound colonisation (treated by wound exposure, washing and topical nanocrystalline silver dressings), as well as enterococcal bloodstream sepsis requiring intravenous antibiotics. On day 111, the patient was discharged to in-home rehabilitation with outpatient monitoring of stage IV chronic kidney disease and return of lower limb sensation (Figure 3). He was able to independently attend to all activities of daily living, including outdoor mobilisation.
Discussion
Biodegradable temporising matrix providing a temporising scaffold and immediate coverage in preparation for staged skin grafting is a useful rung on the reconstructive ladder. Successful use has been described in major burns, necrotising fasciitis and degloving injuries.5–8
Immediate coverage negates reliance on fragile, undulated granulation tissue to bridge over ‘ungraftable’ beds. While VAC® therapy dressings (KCI Medical Australia Pty Ltd, Level 7/15 Orion Road, Lane Cove West NSW 2066, Australia) alone would promote granulation tissue formation, in the setting of such extensive geographical wounds and Pseudomonas colonisation, successful coverage would have been improbable. VAC® dressings are never used by the authors to promote capillary ingrowth into the BTM, which, without nutrient properties in its synthetic polymer platform, may possess a higher degree of bacterial resistance than biological alternatives such as Integra® (Integra LifeSciences Corporation, 1100 Campus Rd, Princeton, NJ 08540, United States).9 In colonised wounds, antimicrobial dressing changes external to the BTM allow penetration through the factory and hand fenestrations without risking detrimental mechanical debridement of the fragile granulating bed. Using this method, total BTM integration was accomplished resulting in a degree of graft take that would probably be unachievable without a dermal substitute considering the wound complexity, surface area and colonisation.
The drilling of burr-holes in bone denuded of periosteum provides a route for transcortical vascular ingrowth into the BTM that would otherwise rest on a dry, avascular bed and be solely reliant on peripheral ingrowth. Acknowledging the potential for contiguous bacterial spread, this technique has been used by one author in deep burns and adapted for exposed calvarium without complicating osteomyelitis.10
This novel indication mirrors the well-documented fibroblastic ingrowth, dermal collagen deposition and neovascularisation seen in burn wounds prior to the delamination of the superficial, non-biodegradable, polyurethane layer and describes the surgical techniques necessary to optimise those processes in a complex pathology with challenging wounds.
Conclusion
Limb salvage after extensive soft-tissue loss in the setting of critical illness is infrequently considered, and amputation is expeditiously performed. This report documents the authors’ first experience of BTM as a resource-sparing alternative to free-tissue reconstruction to avoid bilateral lower limb amputation in the setting of extensive myonecrosis following meningococcal B infection.
Patient consent
Patients/guardians have given informed consent to the publication of images and/or data.
Conflict of interest
Authors disclose small shareholdings in PolyNovo Biomaterials Pty Ltd. No authors have current affiliations with PolyNovo Biomaterials Pty Ltd. PolyNovo Biomaterials Pty Ltd had no input in the design, writing or editing of this manuscript.
Funding declaration
The authors received no financial support for the research, authorship, and/or publication of this article.
Revised: August 12, 2020 AEST