Introduction
The goal of laryngeal reconstruction is to maintain a patent airway and provide a relatively normal voice. The most common technique to reconstruct the larynx or hemilarynx is to wrap a harvested flap around, or attach a flap to, donor cartilage from a different site. These reconstructions can be quite complex and multistage with potential donor site morbidity and without transfer of the same tissue type. Our alternative solution to this relatively complex surgery is to use a pedicled facial artery myomucosal (FAMM) flap which provides both normal mucosal lining and sufficient bulk to avoid the use of a cartilage graft. With the exception of a canine model,1 the use of the FAMM flap has not been reported as having been used in hypopharyngeal or laryngeal defects.
Case report
A 57-year-old otherwise healthy man presented to his general practitioner (GP) after noticing a lump on his neck while shaving. Ultrasound, CT, MRI, positron emission tomography (PET) scan and biopsy were performed, diagnosing a low-grade chondrosarcoma of the right thyroid cartilage. He was seen in a head and neck multidisciplinary clinic (video available as a supplementary file) and a two-part surgery was planned to include a vertical hemi-laryngectomy with tracheostomy by ear, nose and throat (ENT) surgeons, followed by reconstruction by plastic and reconstructive surgeons. Reconstructive techniques considered included deep circumflex iliac artery (DCIA) inner cortex flap, radial artery forearm flap (RAFF) with bone or pedicled FAMM flap ± cartilage.
Following tracheostomy, the complete right thyroid cartilage was resected with a 5 mm margin taken on the contralateral side. The cartilage and cricothyroid membrane were divided, and the left anterior cord was sectioned allowing removal of the right hemilarynx. The arytenoid and the superior aspect of the thyroid lamina were cleared and the epiglottis was divided before the specimen was removed. Bilateral arytenoids, cricoid and epiglottis were preserved. The left vocal cord was attached to the thyroid lamina.
For reconstruction, a 2.5 × 5.0 cm anterograde pedicled FAMM flap was harvested from the right buccal mucosa. The flap, including the mucosa and buccinator muscle, was then islanded on the facial artery and its venae comitantes. A separate incision over the mandibular edge was made to help with pedicle dissection. The facial artery was dissected below the mandible until adequate pedicle length was achieved, but did not require dissection deep to the submandibular gland or behind the digastric muscle. The flap was then tunnelled under the platysma into the defect, then sutured to the residual left thyroid cartilage and surrounding soft tissue, thus using tissue bulk rather than structural cartilage for reconstruction. The flap was inset and the strap muscles were repaired, covering the FAMM flap.
The patient returned to theatre on postoperative day 1 for evacuation of a serous collection in the neck due to a blocked drain, with no evidence of salivary leak. He then healed well with no further complications.
On six-week postoperative review the patient was eating and swallowing well, had no restriction of mouth opening and speech was intelligible but reduced in volume.
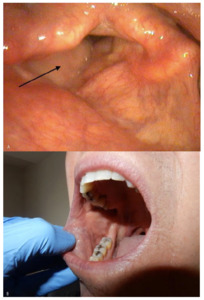
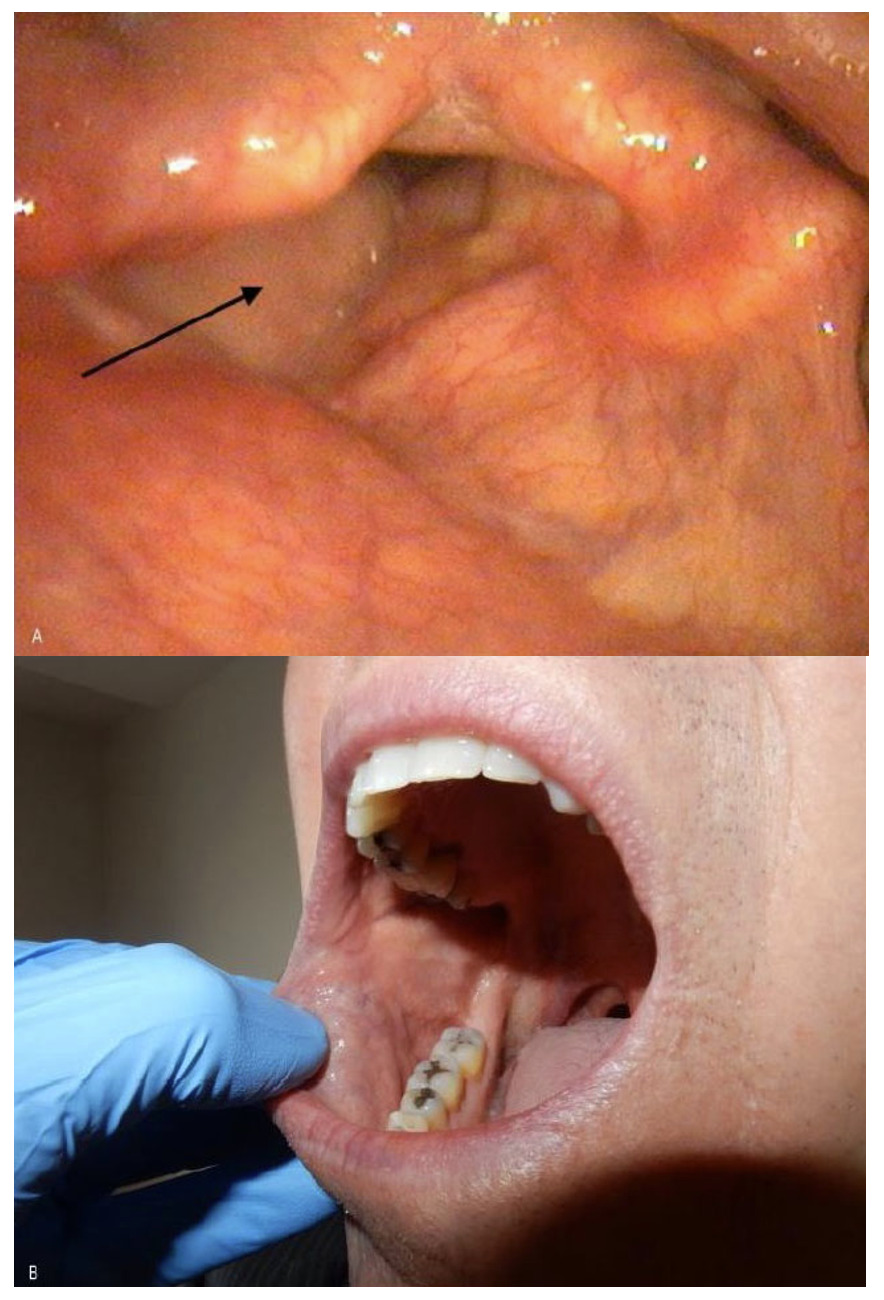
On five-year postoperative endolaryngeal examination, the flap looked like normal mucosa and a patent airway was maintained (Figure 1a). There were no donor site issues (Figure 1b) and speech had further improved (video available as a supplementary file).
Discussion
Myriad reconstructive techniques have been described for this difficult defect, which implies that no technique is perfect. Some examples include: a free temporoparietal fascial flap based on the superficial temporal artery sutured to a buccal mucosa graft and cartilaginous thyroid graft,2 a pectoralis major myocutaneous flap3 and a radial forearm flap wrapped around a segment of cervical trachea.4 Numerous types of cartilage are used such as auricular5 and nasal septal cartilage.6 These techniques all come with inherent complexities and donor site issues.
The FAMM flap, introduced by Pribaz and colleagues in 1992,7 has become increasingly popular over the past decade. Based on the facial artery, it consists of a portion of the buccinator muscle and buccal mucosa. Previously FAMM flaps have primarily been indicated for oral and oropharyngeal use and the majority of the literature pertains to this.
Facial artery myomucosal flaps offer many benefits and few disadvantages, as summarised in a 2015 systematic review by Ayad and Xie.8 Many of the advantages of a FAMM flap applied to our patient including ease of harvesting, large axis of rotation and generous and adjustable length for more distant reconstruction sites without being under tension.9 The flap is thin and pliable, which allows it to be tunnelled under the platysma. It is composed of mucosal tissue, thus ideal for replacing the luminal mucosa of the larynx,9 compared to flaps and grafts that have potential drawbacks due to histological differences such as reduced sensitivity and potential hair growth.8–10 As the FAMM flap has enough intrinsic bulk, donor cartilage was not needed to provide structural rigidity. Primary closure was able to be performed at the donor site. An external scar can often be avoided with this flap, and its strong vasculature can better withstand postoperative radiotherapy.5,6
There are few disadvantages of a FAMM flap. It is limited by width, which was not an issue with this patient.5,6 It sometimes requires a two-stage procedure, which can be overcome by completely islanding the flap and tunneling it into the neck. One of the drawbacks of the literature pertaining to FAMM flaps is the low level of evidence, being comprised mostly of small-scale studies and case reports.
Conclusion
This relatively simple technique used for the reconstruction of the hemilarynx provided a novel solution with excellent functional results and no donor site morbidity. The return to normal voice and swallowing function warrants the consideration of the FAMM flap in the setting of perilaryngeal defects.
Supplementary online material
https://youtube.com/playlist?list=PLz13kz4bLadS4QamFDrvyd7a-Y3U-JhCt
Patient consent
Patients/guardians have given informed consent to the publication of images and/or data.
Disclosure
The authors have no financial or commercial conflicts of interest to disclose.
Funding declaration
The authors received no financial support for the research, authorship, and/or publication of this article.
Revised: 2021 May 5 AEST