Introduction
Necrotising soft tissue infection (NSTI) is a rapidly progressing disease that presents a surgical emergency. Timely antibiotics, radical debridement of infected tissues and adjuvant hyperbaric oxygen therapy are the foundations of its treatment. With improved recognition and management of NSTI in recent years, mortality rates have declined but patients are often left with large, morbid soft tissue defects.1 Split-skin graft (SSG) is the main reconstruction technique due to its simplicity and dependability. To graft a wound bed requires a blood supply, minimal bioburden and sufficient drainage of potential collections.1 However, where there is a contaminated wound with extensive soft tissue loss and exposed tendons, SSGs are at risk of failure. Further, SSGs do not properly replace the lost dermis and are prone to rigidly adhere to underlying structures.2,3
Dermal substitutes, as well as creating a suitable wound bed for grafting, aim to recreate the inherent thickness and pliability of skin.4 One innovation, Novosorb™ (produced by PolyNovo Ltd, Port Melbourne, Australia), is a biodegradable temporising matrix (BTM) that is an entirely synthetic implantable dermal matrix that creates a neo-dermis in complex wounds. Biodegradable temporising matrix use requires a two-stage process: first, implantation of the synthetic bilaminar matrix and allowing for host integration; and second, delamination of the superficial layer and application of a SSG to the remaining vascularised matrix.2,3
Case
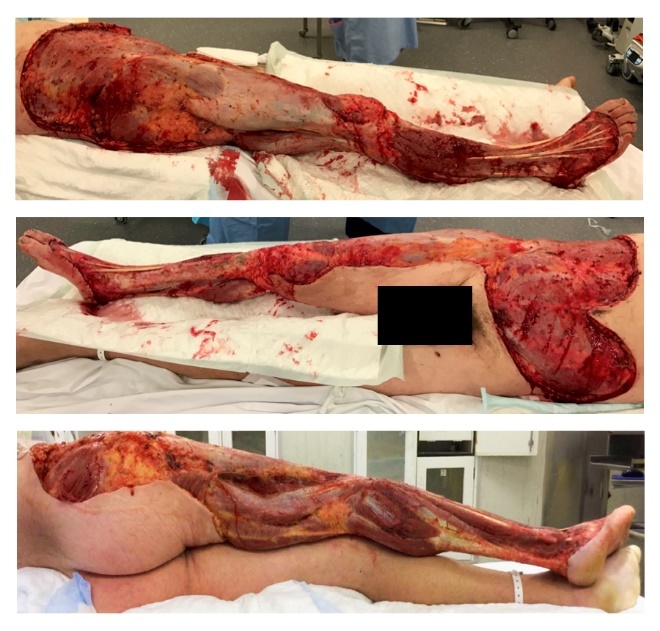
A 42-year-old male with septic shock secondary to NSTI of the right leg and arm was transferred to a tertiary trauma hospital for further management and adjuvant hyperbaric oxygen therapy. The patient was an otherwise healthy individual and avid surfer who had sustained abrasions to his right leg and arm from coral while surfing in Bali four days prior to presentation. After four extensive debridements, the total soft tissue loss to his anterior abdomen, right thigh, right circumferential leg, dorsum of the right foot and right arm (Figure 1) was 32 per cent of body surface area.
At day 17 post-admission, the patient’s wounds appeared clean and cultures were negative. Reconstruction began with cadaveric allografts as a temporary dressing, so as to improve and prepare granulation tissue. These allografts, however, became infected with Pseudomonas aeruginosa. Further serial debridement of the infected devitalised tissue led to the right leg anterior compartment tendons becoming exposed, desiccated and lacking paratenon—hence this portion of the wound was no longer graftable. Free tissue transfer was decided against due to the infection, the large size of the soft tissue defect and the presence of suboptimal recipient vessels within the zone of infection.
Novosorb BTM was at the time undergoing a trial at our institution for the management of extensive burns. It was decided to use this dermal substitute to create a graftable bed in the right leg and dorsum of the foot, specifically targeting the exposed tendons. Approval from the Therapeutic Goods Administration of Australia was obtained for its use in this case.
At day 37 post-admission, the right leg and dorsum of the foot were surgically prepared and implanted with Novosorb BTM. Current protocols require implantation of BTM onto a clean non-infected wound prepared with betadine. The BTM is secured with quilting staples and dressed with Acticoat™ (Smith & Nephew, London, UK).
As recommended, the patient underwent regular dressing changes while the implanted BTM became integrated over the next 21 days, which allowed for monitoring of formation haematoma and infection. Over this period, the BTM became gradually more hyperaemic and vascularised (Figure 2). Twenty-four days after implantation the BTM’s surface sealing layer was ‘delaminated’, revealing a vascularised matrix. The right leg anterior compartment tendons were no longer exposed. The wounds were then reconstructed using a 2:1 meshed SSG.
Eighty-three days post-admission the patient was discharged to a rehabilitation hospital. The majority of his grafts had healed, apart from small areas of hypergranulation (Figure 3), which resolved with dressings. Twenty months after the final SSG he has achieved 10° of dorsiflexion and 30° of plantarflexion in his right ankle. He is currently walking independently without aids, working full-time in sales and has returned to surfing.
Discussion
It is well understood by plastic and reconstructive surgeons that there are shortcomings in the techniques used to reconstruct extensive, full-thickness skin defects. Presently, no reconstruction technique fully replaces the critical qualities of the lost skin. Split skin grafts are currently favoured in reconstruction due to their technical simplicity, reliability and the presence of abundant donor sites.3,4 However, SSGs do have limitations. As adenexal structures are not transported in grafting, SSGs are unable to auto-hydrate—resulting in itching, dryness and cracking and leaving grafted areas prone to microtrauma and infection. As the suppleness and mobility of skin is owed to its dermis, the reduced thickness of elastic dermis in a SSG and its inherent tendency to adhere to underlying structures can result in the restriction of joints and tethering of tendons.2,3,5
Exposed tendons in the presence of infection and where there is large TBSA soft tissue loss pose a reconstructive dilemma.1,6,7 When faced with the problem of preserving exposed tendons in a wound where skin grafting alone will not suffice, there is a lack of local tissue transfer options and any free tissue transfer is both technically challenging and at risk of failure.7–9 Contributing to this difficulty are the minimal potential donor sites for a large defect and the condition of vessels lying within the zone of injury—microvascular anastomosis is at risk of failure due to friable vessels, the presence of microthrombi and an inability to cover the pedicle.7,9 Alternative treatment options are: sacrificing function and excising the tendons; employing negative pressure wound therapy (NPWT); or utilizing a dermal matrix.6,7,9 Negative pressure wound therapy can be employed until a graftable wound-bed forms, however due to the large soft tissue defect this would require a prolonged period and is at risk of failure.7
Dermal substitutes have been tested with varying degrees of success to address the limitations of SSGs. By providing a scaffold that promotes new tissue growth, dermal substitutes tend to recreate some of the skin’s inherent thickness and pliability. The formation of a neo-dermis also tends to resist secondary contracture of wounds.2–5 When combined with an impermeable seal, dermal substitutes provide a temporary barrier that slows fluid loss and deters pathogens. The ideal dermal matrix should demonstrate long-term biocompatibility, including abilities for host cell infiltration, neovascularisation, deposition of endogenous collagen, and a minimal immune response while still being biodegradable.4,5
Integra® (Life Sciences Corp, Plainsboro, USA) is a commonly used dermal substitute comprised of purified bovine tendon collagen cross-linked with shark cartilage glycosaminoglycan and sealed with a non-porous silicone layer.4,5 Integra succeeds in forming a neo-dermis while biodegrading and its use in NSTI has been advocated.6,7 Nevertheless, its high cost, vulnerability to infection and induced host response that leads to extrusion are all causes for concern.4,5
NovoSorb™ BTM is a biodegradable polyurethane foam sealed with a removable non-biodegradable polyurethane outer layer.2–5,10 As it is a synthetic dermal template it avoids the risk of immunogenic rejection and the potential disease transmission involved in animal- or human-based products.2–5 In preliminary studies on animals and humans, BTM supported the ingrowth of host fibroblasts, deposition of dermal collagen matrix and neovascularisation.2–5,10 It also allowed the migration of keratinocytes from the wound edge and basement membrane formation.2,3 In in-vivo studies, it did not induce a local or systemic cytotoxic response and it appears to integrate in spite of wound colonisation with pathogenic organisms.5 When compared with Integra, NovoSorb™ BTM exhibits less wound contraction, a greater local inflammatory response and a more extensive vascular network.4,5
Biodegradable temporising matrix has undergone trials for burns, small wounds, free flap donor sites and a single reported case of anterior cervical NSTI.4,5,9 The current protocol requiring close monitoring after implantation while it integrates is potentially resource intensive—a period of three to four weeks is required for the neo-dermis to form and integrate with the polyurethane foam matrix.3,5 Once integrated, the BTM is delaminated and the underlying vascularised matrix reconstructed with a SSG.3,5 Thus BTM requires a two-staged procedure.2–5,10 The polyurethane foam matrix completely degrades and resorbs in approximately six months.2,3
Conclusion
Necrotising soft tissue infection is a devastating disease that often leaves a patient with large full-thickness skin loss. SSGs provide a reliable and functional reconstruction but have numerous shortcomings. The synthetic dermal template NovoSorb™ is a product that offers a two-staged reconstruction for complex skin defects. We demonstrated its successful application in treating a large soft tissue defect that contained exposed tendons and presented a high risk of infection. It has the potential to offer a thicker, more durable and more mobile skin reconstruction when compared with SSG alone.
Patient consent
Patients/guardians have given informed consent to the publication of images and/or data.
Disclosure
Ms Heather Cleland is conducting a separate clinical trial that is funded by Polynovo, manufacturer and marketer of NovoSorb™ biodegradable temporising matrix.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Prior publication
The case in this manuscript was presented as a poster in the 2017 Plastic Surgery Congress of Australian Society of Plastic Surgeons on the Gold Coast, Australia.
Revised: September 16, 2018 AEST

_day_5_post_implantation_(b)_day_21_post_imp.jpeg)

_day_5_post_implantation_(b)_day_21_post_imp.jpeg)
